ZINGA can easily be removed using Zingasolv. Also dried ZINGA can be redissolved using Zingasolv.
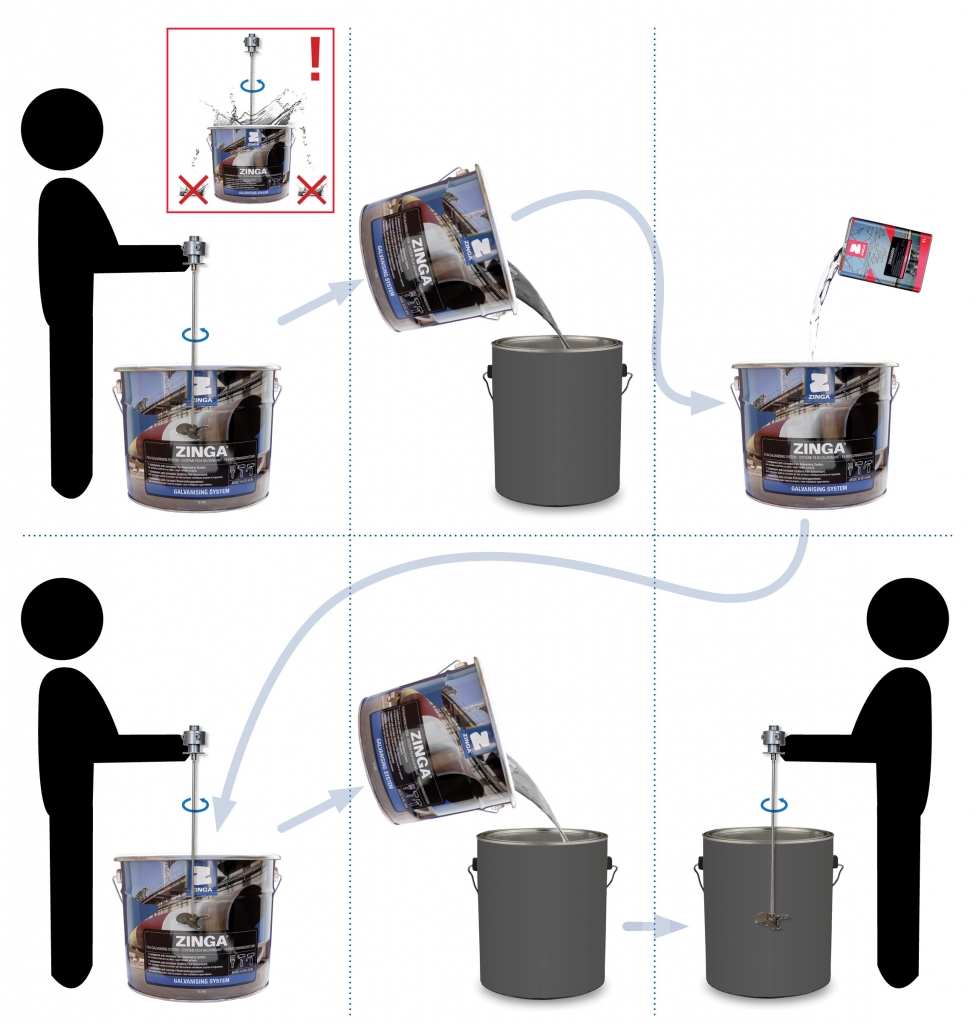
ZINGA should be stirred using mechanical tools.
Insert a mechanical mixer into the pot and stir for at least 2 minutes for small quantities (1 – 2 kg) and at least 5 minutes for large quantities (5 – 25 kg).
To test correct stirring, perform one of two options:
- Weigh a measured volume: if the calculated density (weight divided by volume) is between 2,58 and 2,73 kg/L, the product is adequately stirred.
- Empty the jar after stirring in another jar. Dilute the remaining product with Zingasolv. Mix the diluted product. Poor this in the new jar. Mix the new jar thoroughly.
ZINGA can be overcoated, but is -like any Zinc rich coating- sensitive to solvents. Therefore compatibility must be ensured. In general, fast-curing low solvent containing topcoats perform better. To test, apply a small amount of topcoat on a non-critical, non-visible part of the structure which has been coated with ZINGA and observe if any reaction occurs. To avoid any problems, we advise the use of a sealer (see: sealers on ZINGA).
No, ZINGA should be thoroughly stirred before application. It is very important that the ZINGA is a homogeneous product during application to ensure cathodic protection of the structure.
Zingametall is the manufacturer of ZINGA which can be used to treat metallic structures to prevent them from rusting.
Zingametall has contacts with applicators who have experience in using ZINGA, but does not offer applications in its facilities.
No, ZINGA is only available in grey.
The grey is attributed to the extremely high Zinc content of the coating. In order to have enough coloured pigment in a paint, the Zinc content is dropped below the level required for galvanic protection.
The performance of ZINGA is, like any coating or paint, highly dependent on the degree of surface preparation before application. Therefore, ZINGA can be applied on a slightly rusted surface, but due to the barrier of rust between ZINGA and steel, the galvanic protection is affected and therefore the performance of ZINGA is reduced.
Since ZINGA can offer a sustainable, longlasting protection of steel surfaces, it is profitable to invest in a thorough surface preparation which prevents premature failure of the coating.
Yes, the welding of steel coated with ZINGA (max. 60 μm DFT) is possible without excessive zinc fumes because the heat of the approaching weld bead burns off the organic binder well below the melting point of zinc.
The remaining zinc dust is removed from the weld zone by convection leaving the weld-area free from contamination. If the applied DFT is more than 60 μm, the excessive coating must be removed with a wire brush. The zinganised steelwork must be free of oil, grease and any chemicals that are flammable. Dust and debris should be removed by air gun or by vacuuming.
Do not use silicone based anti-spatter spray on ZINGA because this will cause adhesion failure of any subsequent coatings that will be applied on the ZINGA. Please ensure that adequate ventilation and extraction is used at all times. When welding zinganised steel, the operator must wear a mask that conforms to the regulations.
ZINGA can be used on welds to ensure a cathodic protection over the entire structure. Before accepting the job, please verify that the welds are in good condition. Holes, undercuts or cracks should be remedied by welding or grinding until a uniform surface is obtained.
The surface should be degreased and cleaned to cleanliness degree St 2. Weld spatters must be removed. Eliminate all slag and other surface imperfections with a mechanical needle hammer. Before the application of ZINGA, the welding seams and the surrounding surface must be roughened: preferably by grit-blasting, or otherwise with a bristle blaster, a needle hammer or with rough sand paper (60 to 80). Be careful not to polish the surface!
After roughening of the surface, a thorough dedusting of the areas to be treated is vital. Apply the ZINGA by brush in overlap of minimum 5 cm, in one or more layers in order to obtain the specified DFT.
ZINGA has been developped to protect ferrous structures.
The performance of ZINGA is highly dependent on the degree of surface preparation. How better the preparation, how better its performance. Please see surface preparation for detailed explanation.